Therefore, more gaseous molecule means more randomness, higher entropy. 
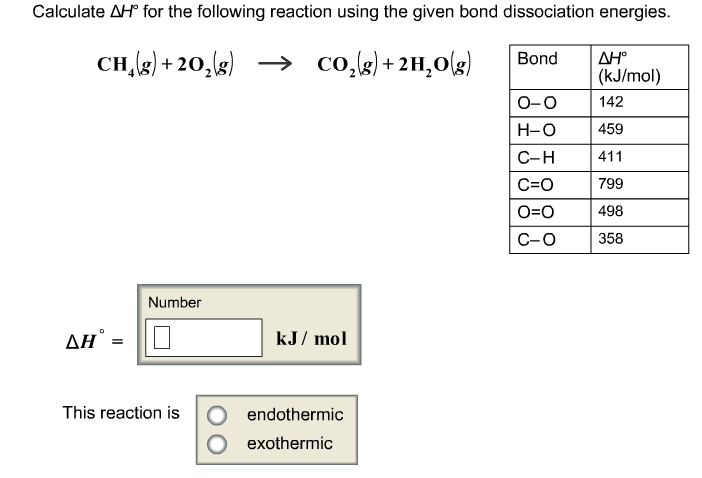
It can be observed that the total number of moles of the product side is greater than the total number of moles of the reactant side. So Hess's Law tells us that delta H of this reaction. The chemical species present are all gaseous compound. The free energy change of a reaction is a mathematical combination of the enthalpy change and the entropy change. We know how much energy it takes to go from the elemental form to C and D. First, write the reaction for the combustion of C 2 H 6 C_2H_6 C 2 H 6 and balance the reaction.Ģ C 2 H 6 ( g ) 7 O 2 ( g ) → 4 C O 2 ( g ) 6 H 2 O ( g ) 2 \ C_2H_6 (g) 7 \ O_2 (g) \rightarrow 4 \ CO_2 (g) 6 \ H_2O(g)Ģ C 2 H 6 ( g ) 7 O 2 ( g ) → 4 C O 2 ( g ) 6 H 2 O ( g )
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
